题文
已知热化学方程式:
H2O(g) = H2(g) +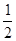 O2(g) △H =" +241.8" kJ·mol-1
O2(g) △H =" +241.8" kJ·mol-1
H2(g)+ 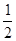 O2(g) = H2O(1) △H = -285.8 kJ·mol-1
O2(g) = H2O(1) △H = -285.8 kJ·mol-1
当18g液态水变为水蒸气时,其热量变化是
| A.吸热44 k J | B.吸热88kJ | C.放热44kJ | D.吸热2.44 kJ |
已知热化学方程式:
H2O(g) = H2(g) +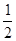 O2(g) △H =" +241.8" kJ·mol-1
O2(g) △H =" +241.8" kJ·mol-1
H2(g)+ 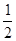 O2(g) = H2O(1) △H = -285.8 kJ·mol-1
O2(g) = H2O(1) △H = -285.8 kJ·mol-1
当18g液态水变为水蒸气时,其热量变化是
| A.吸热44 k J | B.吸热88kJ | C.放热44kJ | D.吸热2.44 kJ |
试题篮
()